Data Integrity
Data Integrity Resources for Pharmaceutical Manufacturing
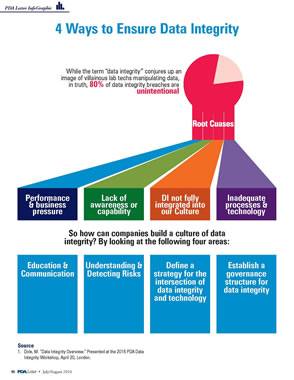
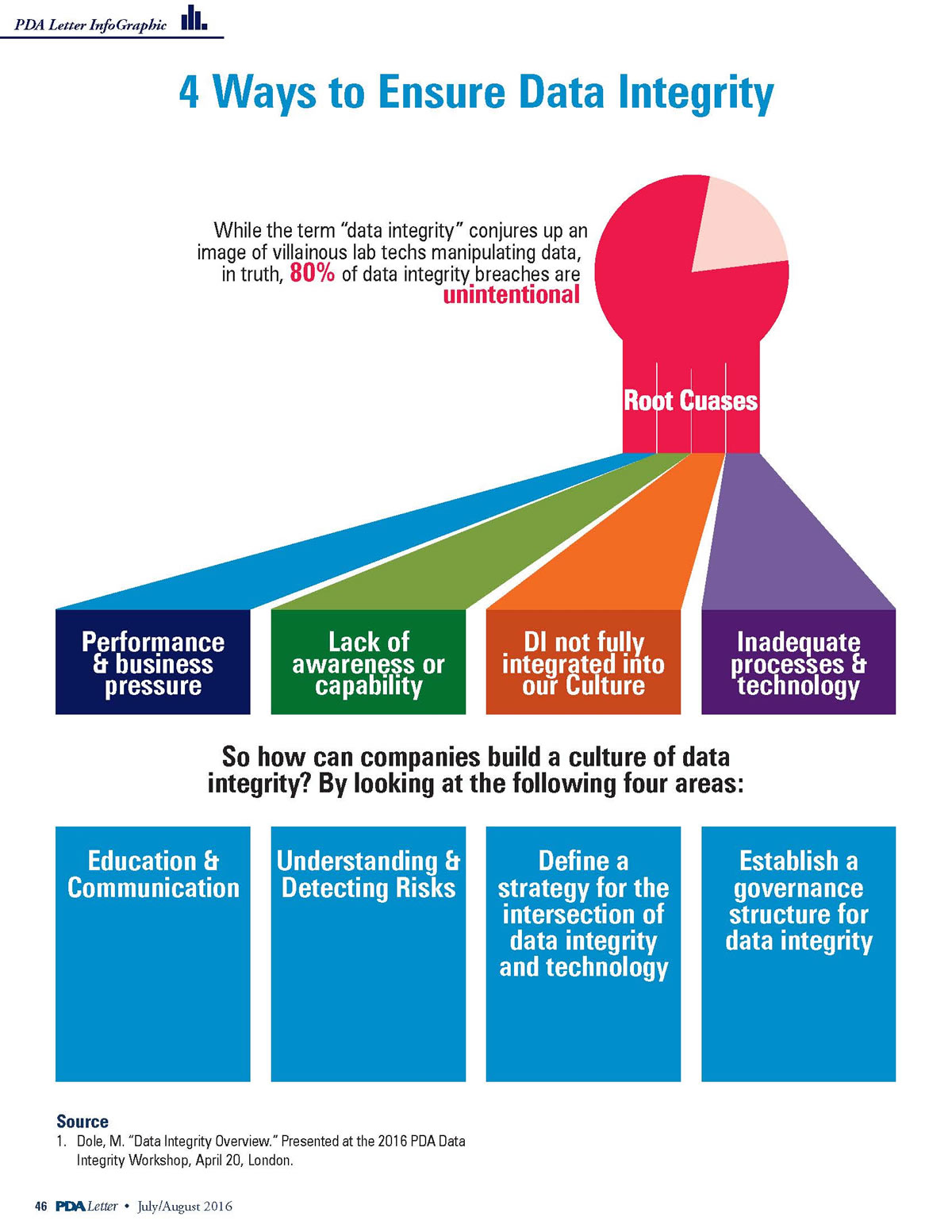
A data integrity program is a significant component of a company's Quality System. It provides foundational assurance that the data the company uses to demonstrate that its products are safe and effective for their intended use are in compliance with regulatory requirements. A data integrity program recognizes the company's responsibility to prove the origin, transmission and content of the company's data are what they are purported to be.
Data integrity has been and currently is a major global concern of Health Authorities and the pharmaceutical industry. Although not a new issue, numerous recent Health Authority enforcement actions, such as Warning Letters, Import Alerts, Product Detentions and suspension or revocation of Marketing Authorizations, have focused attention on data integrity.
To provide resources that holistically address issues related to data integrity, PDA is developing a comprehensive set of tools for industry that includes a Code of Conduct, Technical Reports and Points to Consider documents.
- Promote harmonized standards for compliance with regulatory expectations for maintaining data integrity
- Define mechanisms for detecting non-compliance and outline a clear methodology for remediating gaps
- Serve both industry and regulators by creating and defining solutions for the increasing number of failed inspections where firms lack not only the necessary controls to ensure data integrity but also the expertise to detect and resolve non-compliance
- Develop a methodology for restoring confidence in a system and organization to avoid revenue loss and regulatory impacts
Technical Report Series
PDA's Technical Report (TR) series on data integrity is intended to provide a roadmap for the creation and integration of a Data Integrity Management System as a core element of the QMS. Each TR in the series is intended to focus on a specific element of the QMS and to provide a detailed strategy and framework for establishment of controls and processes necessary to ensure data integrity within that area.
PDA Technical Report No. 84 Integrating Data Integrity Requirements into Manufacturing and Packaging Operations
This TR discusses regulatory trends, risk management concepts, and recommendations for implementing appropriate data integrity controls in manufacturing operations applicable to paper-based, electronic-based, and hybrid systems. The case studies included in this technical report provide examples of how to assess current data integrity risks and implement the concepts presented in the report.
PDA Technical Report No. 80 (TR 80) Data Integrity Management System for Pharmaceutical Laboratories
Gain the framework and tools necessary to establish a robust data integrity management system to ensure data integrity for paper, hybrid, and computerized systems within the laboratory. This TR outlines regulatory requirements and expectations and best industry practices to ensure data integrity, to highlight common gaps in laboratory data management practices, and to recommend methods of remediation.
PDA Points to Consider: Fundamental Concepts for Data Integrity
This document provides a brief overview of the key concepts needed in a program to ensure the integrity of GxP data in pharmaceutical manufacturing operations. It explains fundamental concepts for data integrity prevention, detection and response and defines basic concepts, including ALCOA.
PDA Points to Consider
Best Practices for Document/Data Management and Control and Preparing for Data Integrity Inspections
PDA has identified eleven current questions related to data management and control that have been frequently cited in FDA Inspections or have led to FDA regulatory actions. The purpose of this document is to help to clarify some of these issues for industry and to help facilitate better compliance by sharing PDA members' expertise in and understanding of current best practices
Read morePDA Data Integrity Deliverables
PDA Data Integrity Code of Conduct Impacts Industry
How widespread has been the uptake of PDA’s Elements of a Code of Conduct for Data Integrity in the Pharmaceutical Industry and how has it impacted those companies who have downloaded it?
Read morePublications
Elements of a Code of Conduct for Data Integrity in Pharmaceutical Manufacturing
The Elements of a Code of Conduct for Data Integrity were developed by a team with expertise in the fields of quality, regulatory affairs, auditing and manufacturing and reviewed by attorneys specializing in food, drug and labor law.
Designed to encourage widespread implementation, this document is written for easy adoption, in part or in its entirety, by companies, if they so choose, without the need for extensive rewriting of the document. Therefore, the terms ‘shall' and ‘must' have been used to permit the Code to be enforceable by those companies that choose to adopt it.
This document is intended to reinforce a culture of quality and trust within the pharmaceutical industry. It is not intended to be a regulatory standard or guidance, nor is it intended to supersede any country-specific or local laws and regulations governing labor, privacy and/or employee rights. The Elements of a Code of Conduct for Data Integrity in Pharmaceutical Manufacturing is available for FREE download to the pharmaceutical industry for internal use. The selling of any portion of this Code is not permitted.
PDA Keynote Presentation at Interphex 2018
DI Tech Report Leader, Els Poff, presented an overview of PDA activities and tools to support industry in meeting health authority expectations for data integrity.
Read morePDA/DHI Book: Assuring Data Integrity for Life Sciences
This new book provides a truly global perspective on data integrity and the solutions available to address this serious issue. It includes two main sections: the regulatory and historic background of data integrity, and practical advice on how to prevent or rectify data integrity breaches.
Each chapter is written by renowned, highly experienced subject matter experts in the fields of compliance and data integrity, and includes a "how to" section with practical, implementable advice. Content is up to date with the latest regulations and guidances, making this the most relevant reference source of its kind. Useful checklists and aide memoirs can be customized by the discerning reader. This book should be equally useful for the quality unit professional, operations manager, validation experts and regulators.
The modular structure allows readers to pick chapters of special interest without having to reach the chapters in order. However, given the usefulness and universal application the "nuggets of wisdom" and advice provided, it is anticipated that readers will want to read the publication in its entirety.