New Approach Suggests Continuous Lyophilization is Possible
Conventional batch freeze-drying has long been the mainstay for stabilizing biologic drug products in storage and distribution, but it presents many challenges. An innovative continuous process for freeze-drying has been developed, however, that may offer a view of the future of freeze-drying for biologics.
The market for biologic drug products, like therapeutic proteins and vaccines, continues to grow. The stability of these products, however, is often limited when formulated as an aqueous solution. Water-mediated degradation pathways can lead to decreased potency or even to toxicity of the drug molecule. Freeze-drying (lyophilization) is a commonly applied low-temperature drying process used to improve the stability of these products during storage and distribution (1). Approximately 50% of the biologic drug products approved by regulators are freeze-dried formulations (2,3). Yet, freeze-drying has a long processing time and carries high costs.
Step-by-Step, Batch-by-Batch
Conventional pharmaceutical freeze-drying of unit doses is a batchwise process during which all vials of the same batch are processed through a sequence of consecutive process steps: 1) freezing, 2) primary drying and 3) secondary drying (3). Vials containing the aqueous drug formulation (i.e., unit doses) are loaded onto temperature-controlled shelves in the drying chamber (Figure 1). During the initial freezing stage, most of the water crystallizes to ice while the solutes also crystallize or form a rigid amorphous glass.
For the subsequent primary drying step, ice crystals are removed via sublimation under vacuum, leaving a porous matrix. Energy is supplied to the frozen product to enhance ice sublimation (endothermic process). Finally, during secondary drying, most of the remaining unfrozen water (i.e., water dissolved in the amorphous phase) is removed by diffusion and desorption until the desired residual moisture content of the dried end product is achieved
At the end of the lyophilization process, the aqueous drug formulation is transformed into a solid and rigid dried cake with an increased shelf life. The most important critical quality attributes (CQAs) of the freeze-dried end product are the (i) API state (e.g., protein conformation); (ii) residual moisture content; (iii) freeze-dried product cake appearance and (iv) reconstitution time. After freeze-drying, these CQAs are evaluated for vials selected at random positions of the batch via offline analytical technique
For more than 80 years, pharmaceutical freeze-drying has been conducted using this unchanged batchwise approach. This traditional batch approach, however, is inherently associated with several disadvantages:

- Batch freeze-drying is inefficient and consumes a lot of time and energy with cycle times that can vary from one to as many as seven days
- The huge size of industrial batches leads to scale-up issues; initial development of freeze-drying cycles is performed in lab-scale equipment, and subsequent steps in the development process, beginning with lab-scale to pilot-scale and, finally, to industrial-scale freeze-dryer
- The freezing stage is uncontrolled, leaving a significant impact on consecutive drying steps; this can cause vial-to-vial variability in the sublimation rate within a batch and between batches (4).
- The heat transfer in the freeze-drying chamber is uneven, resulting in a difference in energy transfer to vials located at different positions on the freeze-dryer shelves (for instance, vials situated at the edge of the shelves are exposed to additional radiant heat coming from the warmer surroundings compared to the vials situated in the middle of the shelves, leading to a higher product temperature for these edge vials that is associated with faster drying rates and a higher risk for collapse) (5)
Both the uncontrolled freezing and the uneven heat transfer culminate in different process conditions for each individual vial in the batch, leading to uncontrolled vial-to-vial and batch-to-batch end product variability. Yet product quality is only determined by testing a limited fraction of vials before releasing the complete batch.
A Fresh Look at Freeze-Drying
To overcome the disadvantages associated with conventional batch freeze-drying, an innovative continuous and controlled freeze-drying concept for unit doses has been developed (6,7). In this approach, all unit operations are integrated in a single production line with continuous feeding of raw materials and the concomitant removal of finished products. This manufacturing approach offers several advantages—avoidance of scale-up issues, reduction in cycle times, lowered production costs, smaller manufacturing installations and improved product quality (process uniformity).
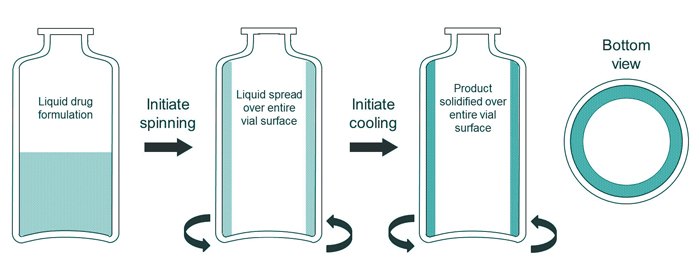
At the start of the continuous freeze-drying process, sterile glass vials are aseptically filled with the aqueous drug formulation before they are transferred to the freezing unit. In the freezing unit, the vials are gripped at their cylindrical walls and rapidly rotated along their longitudinal axis to form a thin layer of product which is spread over the entire inner vial wall (i.e., spin-freezing, see Figure 2). When a homogeneous product layer is obtained, the flow of a cold, inert and sterile gas leads to the cooling and freezing of the solution. Both the temperature and the flow of the gas can be adapted to obtain a specific cooling regime, varying from very fast to slow cooling.
At the end of the spin-freezing step, the product solidifies over the entire inner vial wall resulting in a thin product layer of a uniform thickness (a large surface area and thin product layer). Annealing can be performed by transferring the vials to a chamber with a controlled temperature.
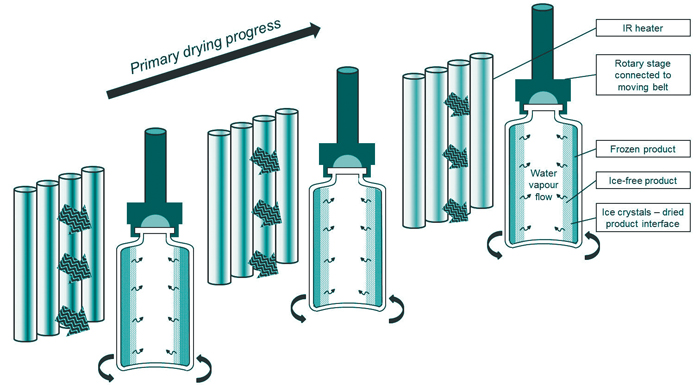
An appropriate load-lock system is used to rapidly transfer the spin-frozen vials between the continuous freezing and the continuous drying units without disturbing the specific conditions of pressure and temperature in each chamber, thereby, guaranteeing the continuity of the process. In the drying chamber, an endless belt system allows the transport of the spin-frozen vials in front of individually controlled radiators which provide a uniform and adequate heat transfer to the entire vial surface to achieve efficient and homogeneous drying behavior (Figure 3). This vial transport takes place in discrete steps. Each vial is rotating very slowly in front of a single radiator, hence, allowing for individual temperature-regulation which enables an optimal drying trajectory for each spin-frozen vial.
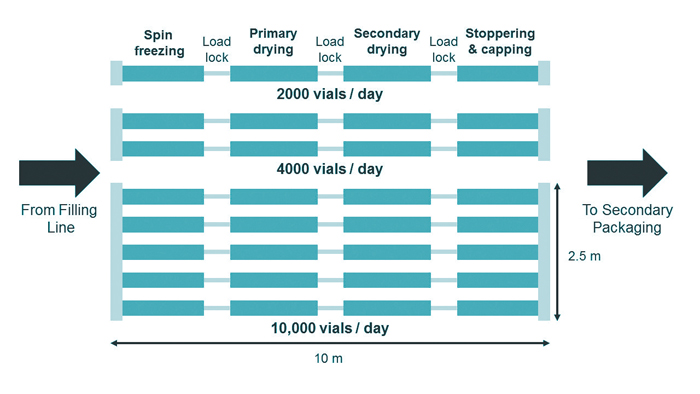
In a conventional freeze-dryer, sublimated ice and desorbed water is collected using cryogenic ice condensers. In the new continuous approach, an appropriate condenser system is used to continuously remove the condensed water. The vial throughput can be increased simply by adding parallel lines in the continuous freeze-drying technology modules, as schematically shown in Figure 4.
Many Benefits to Being Continuous
The continuous freeze-drying technology offers the possibility of implementing process analyzers that allow real-time measurement and control of critical process parameters at the level of the individual vial. Several process analytical tools (PAT), such as near-infrared (NIR) spectroscopy and thermal imaging were evaluated and considered promising. Both techniques also proved highly complementary. NIR spectroscopy can provide detailed in-line information about several quality attributes like residual moisture content, protein conformation or the solid state of different components.
In turn, infrared (IR) thermography allows contactless, real-time and spatial monitoring of the product temperature at the sublimation interface, an essential parameter regarding the cake appearance of the end product.
This integrated approach strongly reduces the variability of CQAs and consistently guarantees the predefined quality of the end product. Hence, the continuous technology meets recent quality-by-design and PAT guidelines issued by global regulators, as opposed to the conventional batch freeze-drying process.
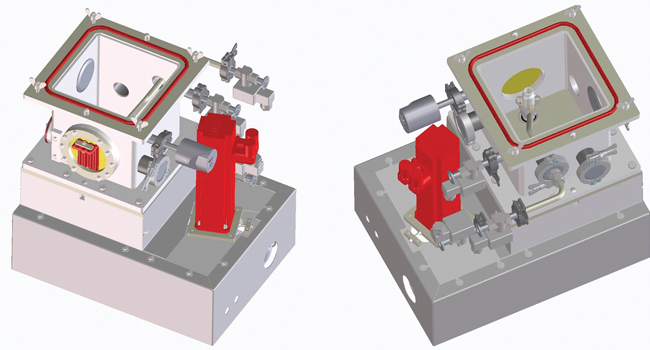
Currently, two different types of continuous freeze-drying prototypes have been built: a single-vial prototype and a GMP-like engineering prototype. The single-vial prototype allows the imitation of the continuous freeze-drying process for one single vial, as identical process conditions can be obtained similar to industrial-scale continuous freeze-dryers (Figure 5).
In case only a very limited amount of drug product material is available, such as at an early stage during development, this prototype allows initial development and optimization of the process and (drug) formulation. In addition, the ability to process small amounts of product makes the single-vial prototype suitable for producing personalized medicines. From a commercial point of view, the single-vial prototype is the perfect tool for R&D laboratories within pharmaceutical companies to gain experience regarding the continuous freeze-drying process.
In the GMP-like engineering prototype, all process modules are integrated, and freeze-drying is executed in continuous fashion (Figure 6). This prototype is engineered around the implementation of needs to create and keep a sterile environment by choosing the proper materials and design principles.
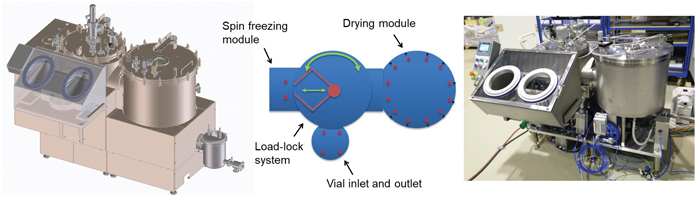
Both prototypes allow the implementation of relevant PATs, including, NIR spectroscopy and thermal imaging, and the practical implementation of mechanistic models leading to optimal process conditions via individual temperatureregulation of IR heaters(7).
Biologic products will play an ever-increasing role in the future of healthcare. And the freeze-drying for these products will be ever more crucial. The two continuous prototypes described here suggest new possibilities to ensure adequate freeze-drying beyond the traditional batchwise process.
References
- Pikal, M.J. “Freeze Drying.” In Encyclopedia of Pharmaceutical Technology, edited by James Swarbrick, James C. Boylan, and Marcel Dekker. 2nd Edition. London, Informa Healthcare, 2002.
- Burns, L. “The biopharmaceutical sector’s impact on the US economy: analysis at the national, state and local levels.” Bioscience Business Roundtable 2009
- Gieseler, H. Insights in lyophilization 2012. Current best practices & research trends. Antwerp, 2012.
- Kasper J., and Friess, W. “The freezing step in lyophilization: physico-chemical fundamentals, freezing methods and consequences on process performance and quality attributes of biopharmaceuticals.” European Journal of Pharmaceutics & Biopharmaceutics 78 (2017): 248–263.
- Van Bockstal, P.J., et al. “Quantitative risk assessment via uncertainty analysis in combination with error propagation for the determination of the dynamic Design Space of the primary drying step during freeze-drying.” European Journal of Pharmaceutics & Biopharmaceutic 78 (2017): 32–41.
- Van Bockstal, P.J., et al. “Noncontact infraredmediated heat transfer during continuous freeze-drying of unit doses.” Journal of Pharmaceutical Sciences 106 (2017): 71–82.
- Van Bockstal, P.J., et al. “Mechanistic modelling of infrared mediated energy transfer during the primary drying step of a continuous freeze-drying process.” European Journal of Pharmaceutics & Biopharmaceutics 78 (2017): 11–21.


